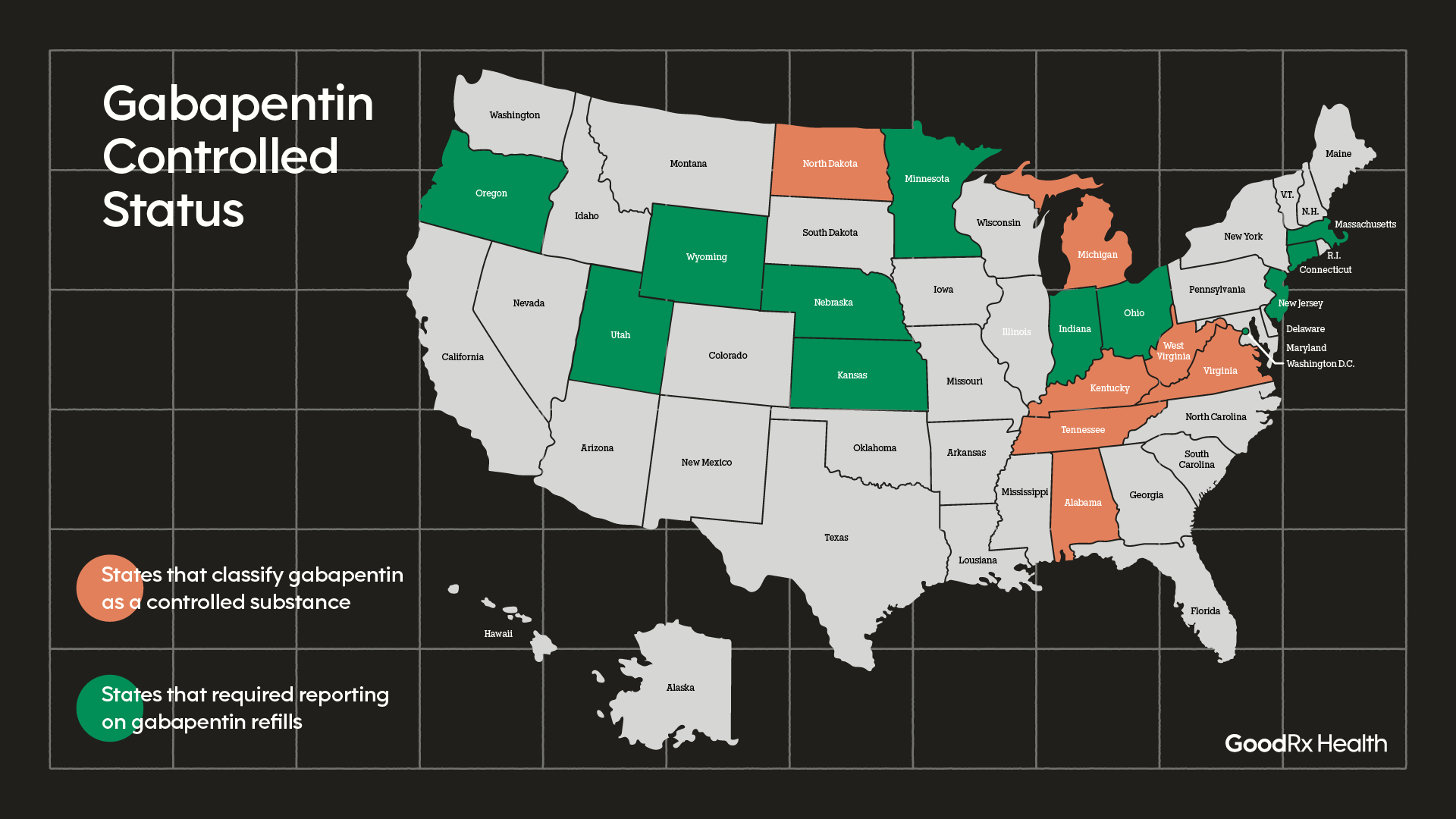
40 explain how controlled substances are identified on their labels
21 USC 802: Definitions (6) The term "controlled substance" means a drug or other substance, ... those terms are defined or used in subtitle E of the Internal Revenue Code of 1986. WHMIS 2015 - Hazard Classes and Categories : OSH Answers Subcategories are identified with a number and a letter (e.g., 1A and 1B). Some hazard classes have only one category (e.g., corrosive to metals), others may have two categories (e.g., carcinogenicity (cancer)) or three categories (e.g., oxidizing liquids). There are a few hazard classes with five or more categories (e.g., organic peroxides).
Drug Classifications, Schedule I, II, III, IV, V - MedShadow Examples of Schedule II controlled drugs include: OxyContin and Percocet (oxycodone), opium, codeine, morphine, hydromorphone (Dilaudid), methadone, Demerol (meperidine), and fentanyl. Examples of Schedule IIN stimulants include: amphetamine (Dexedrine, Adderall), methamphetamine (Desoxyn), and methylphenidate (Ritalin).
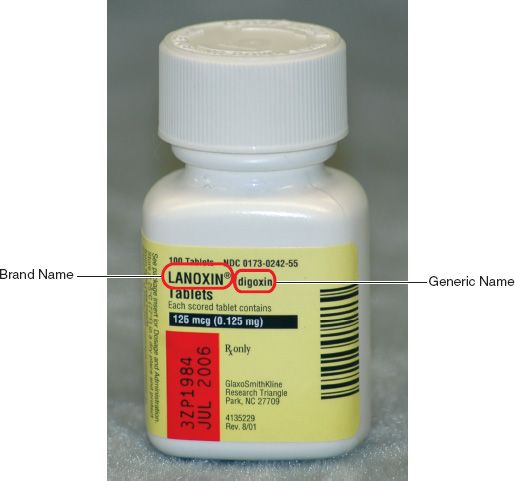
Explain how controlled substances are identified on their labels
4 Controlled Substance Laws and Regulations You Should Know Controlled substance prescriptions have specific requirements. All prescriptions for controlled substances must include the following: 2 Date prescription was issued Prescriber's signature Patient's full name and address Medication name Strength Dosage form Quantity prescribed Directions for use Prescriber's name, address, and registration number. 15.2 Basic Concepts of Administering Medications - Nursing Skills Special Considerations for Administering Controlled Substances. Controlled substances, also called Scheduled Medications, are kept in a locked system and accounted for using a checks and balance system. Removal of a controlled substance from a medication dispensing system must be verified and documented by a second nurse witness. Drug Labeling - StatPearls - NCBI Bookshelf Jun 23, 2022 ... And label and labeling are defined as the following: “(a) ... the content and format of labeling for human prescription drugs and biologics.
Explain how controlled substances are identified on their labels. LABELING AND PACKAGING REQUIREMENTS FOR ... (e) Any term not defined in this section shall have the definition set forth ... print upon the labeling of each controlled substance distributed by him the ... Controlled Substance Schedules - United States Department of Justice Drugs and other substances that are considered controlled substances under the Controlled Substances Act (CSA) are divided into five schedules. An updated and complete list of the schedules is published annually in Title 21 Code of Federal Regulations (C.F.R.) §§1308.11 through 1308.15. Substances are placed in their respective schedules ... Controlled Substances - FAQ - Environmental Health and Safety What are controlled substances? Controlled substances are drugs or chemicals that have the potential to be addictive or habit-forming. The DEA divides controlled substances into 5 categories called Schedules based upon substances' potential for abuse and addictiveness and its' usefulness in medicine. The Drug Scheduling document can be viewed at Definition of controlled substance - NCI Dictionary of Cancer Terms A drug or other substance that is tightly controlled by the government because it may be abused or cause addiction. The control applies to the way the ...
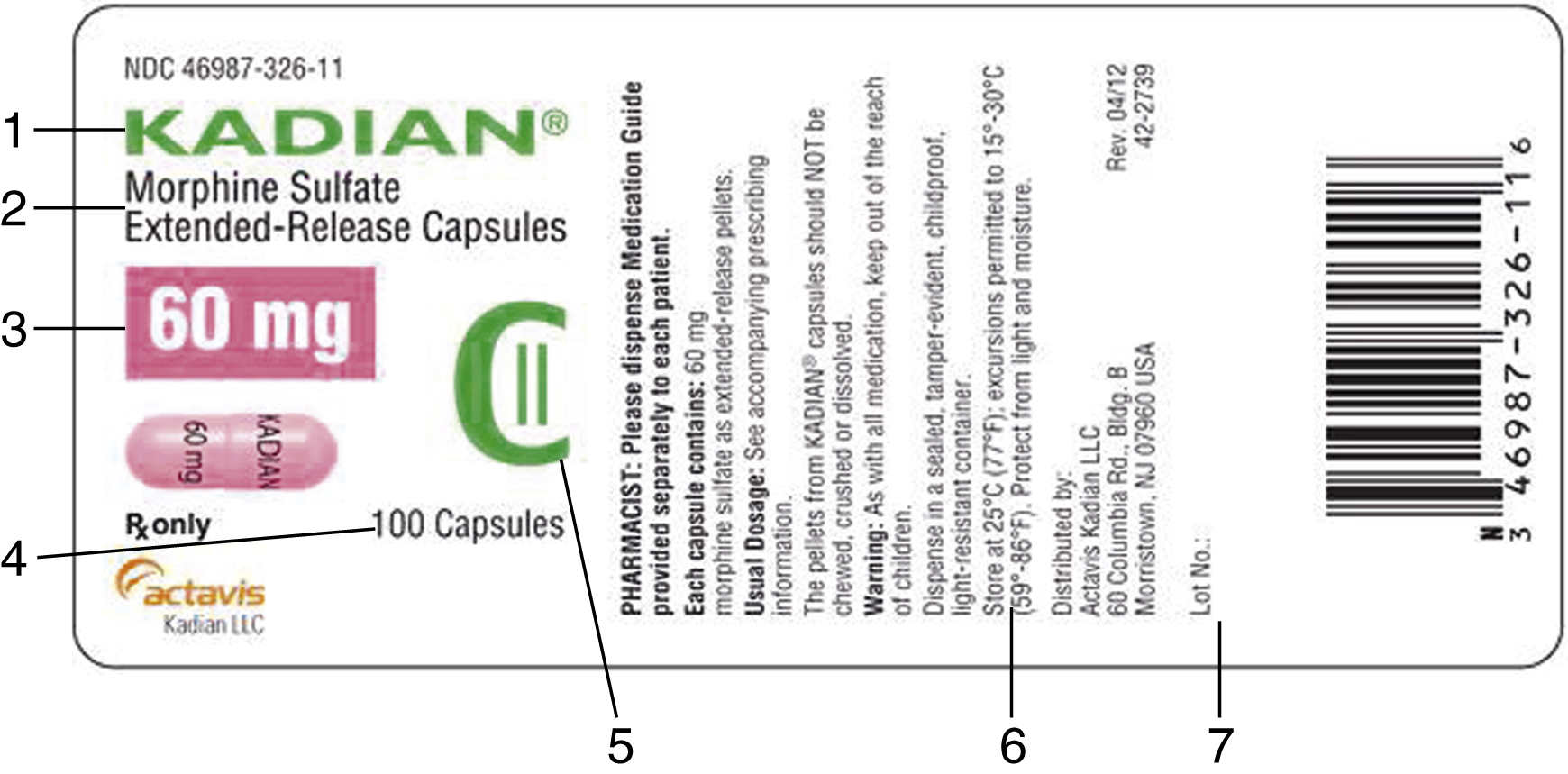
Chapter 51- Principles of Pharmacology Flashcards | Quizlet explain how controlled substances are identified on their labels A large C that means controlled substance, and a roman numeral inside the C that corresponds to the drug's DEA schedule. What are the seven parts of a prescription 21 CFR Part 1302 -- Labeling and Packaging Requirements ... - eCFR (b) Each manufacturer shall print upon the labeling of each controlled substance distributed by him the symbol designating the schedule in which such controlled ... The 5 Types Of Controlled Substances | Chemical Substance Regulation Controlled Substance Schedules. The schedules for controlled substances range from Schedule I to Schedule V. The lower the schedule, the greater the control is. This means that if someone is caught illicitly using, possessing, or selling a schedule 1 substance, the consequences will generally be greater than that of a schedule 5 substance. Food Ingredients & Packaging | FDA FDA maintains educational information, databases and listings related to food allergens, ingredients, food additives, color additives and GRAS substances. Food Additives & Petitions. Generally ...
What Is a Controlled Substance? (DEA Drug Classifications) Controlled substances are drugs that are subject to strict government control because they may cause addiction or be misused. The government's control impacted how these substances are made, used, stored, and transported. Examples of controlled substances include: stimulants opioids hallucinogens anabolic steroids depressants Prescription Labels and Drug Safety - Consumer Reports The U.S. Pharmacopeia, the authority that sets voluntary standards for prescription medication, and the Institute for Safe Medicine Practices recently suggested that it's helpful to patients when... Pharmacy Law and Regulatory - ASHP ACPE activity # 0204-0000-15-443-H03-T. 1 hour (0.1 CEU) This activity will provide an overview of the laws and regulations governing the management of controlled substances in pharmacy practice. The process for utilizing controlled substances, including procurement, dispensing, and disposal will be covered. Strategies for preventing controlled ... Pharmaceutical Labeling 101: FDA Regulations Guide The NDC is a 10-digit code with three segments: a labeler code, a product code, and a packaging code. 2. Makes health data documentation easier Barcodes make it easier to check for lot numbers, expiration dates, and other data digitally. This can protect patients by preventing adverse events. 3. Adds traceability
GHS Hazard Classification: Everything You Need to Know - ERA Environmental T his article is part of ERA's three part series on GHS Hazard Classification.Part one outlines the step-by-step process for classifying your hazardous chemicals. Under the new Globally Harmonized System (GHS) of SDS and Label authoring, chemical manufacturers, importers, and distributors are required to update the way they classify and communicate the hazards of their products.
453 Controlled Substances and Drugs | Postal Explorer - USPS A controlled substance is any anabolic steroid, narcotic, hallucinogenic, stimulant, or depressant drug identified in Schedules I through V of the Controlled Substances Act in 21 U.S.C. 801 and the implementing regulations in 21 CFR 1300.
Prescription of Controlled Substances: Benefits and Risks Controlled substances: These are drugs or medications that possess the potential for being misused and are considered to be substances that have a substantially high risk of resulting in substance use disorder. Opioid analgesics: these are drugs that dull the senses and relieve pain, e.g., morphine. Also, these medications may induce sleep.
21 USC 825: Labeling and packaging It shall be unlawful for the manufacturer of any controlled substance to distribute such substance unless the labeling (as defined in section 321(m) of this ...
Identification, Classification and Labelling of Chemicals ... The packages and containers of dangerous substances and preparations should, in addition to marking only, to have a label with required information. The label should draw attention to the inherent danger to persons handling or using the chemical. Symbols and pictograms have been established for each hazard category listed above.
Drug Scheduling - DEA A Listing of drugs and their schedule are located at Controlled Substance Act (CSA) Scheduling or CSA Scheduling by Alphabetical Order. These lists describes the basic or parent chemical and do not necessarily describe the salts, isomers and salts of isomers, esters, ethers and derivatives which may also be classified as controlled substances.
Controlled substances Flashcards | Quizlet Define controlled substances CS are drugs and other substances that have been determined by federal and state to have potential for creating an addiction or dependency and for leading to various forms of abuse. Many CS are not recognized as drugs. Controlled drugs can be prescription or nonprescription How do federal and state control CS
WHMIS 2015 - Labels : OSH Answers - Canadian Centre for Occupational ... If a signal word is assigned to a hazard class and category, it must be shown on the label, and listed in section 2 (Hazards Identification) of the Safety Data Sheet (SDS). Some hazard classes or categories do not have a signal word assigned to them. What is a hazard statement? Each hazard class and category has an assigned "hazard statement".
Controlled Substances and Dangerous Drugs Dangerous Drugs are defined by the Ohio Board of Pharmacy as any drug or drug product that bears a label containing the symbol “Rx only”, "Caution: Federal ...
Pharmacist Responsibilities for Dispensing Controlled Substances Valid Prescriptions One way pharmacists can protect themselves is by identifying whether a prescription is valid or not before they fill it. The DEA requires that any controlled substance...
Controlled Substances Act of 1970: Definition & History A controlled substance is a medication (or drug or substance) that is regulated by the government, including its possession, manufacturing, and sale. The Controlled Substances Act of 1970 (CSA) was...
Understanding Drug Labels | Basicmedical Key Jun 24, 2016 ... controlled substance A drug or chemical defined by the Comprehensive Drug Abuse Prevention and Control Act of 1970 to have a potential for abuse ...
Chemical Hazards and Toxic Substances - Overview | Occupational Safety ... Most of OSHA's PELs for Shipyard Employment are contained in 1915.1000 - Toxic and Hazardous Substances, and are listed by chemical name. Most of OSHA's PELs for Construction are contained in 1926.55 - Gases, Vapors, Fumes, Dusts, and Mists, and are listed by chemical name. However, many of these limits are outdated.
Drug Scheduling & Classifications (List of Schedule I-V Controlled Drugs) These drugs can be obtained through prescription, but generally are not available over the counter. Schedule IV: Drugs with viable medical use and low probability of use or misuse. Schedule V: Drugs with low potential for abuse (lower than Schedule IV). Heroin LSD Marijuana Ecstasy Quaaludes Bath salts Suboxone Ketamine Anabolic steroids
Prescribing Controlled Substances | Baptist Health CME Identify inpatients who would benefit from non-opioid alternatives and non-pharmacological therapies for pain while maintaining optimal patient care and pain management. Implement best practices in the outpatient setting when prescribing controlled substances and consider alternative therapies.
The Controlled Substances Act - DEA CONTROLLING DRUGS OR OTHER SUBSTANCES THROUGH FORMAL SCHEDULING The CSA also provides a mechanism for substances to be controlled (added to or transferred between schedules) or decontrolled (removed from control). The procedure for these actions is found in Section 201 of the Act (21U.S.C. §811).
Prescribing Controlled Substances | Baptist Health CME Implement best practices in the outpatient setting when prescribing controlled substances and consider alternative therapies. Explain the mechanisms of action that trigger addiction in certain patients and not in others. Identify patients at risk of addiction following opioid treatment for acute pain and implement prevention strategies.
Drug Management - Veterinary Preventive Medicine Define a valid veterinarian-client-patient relationship. Explain rules around prescription of drugs. Briefly explain management of controlled drugs. Describe appropriate disposal of drugs and why this is important. Define and explain veterinary feed directives. This information will be repeated in pharmacology and clinical courses.
Best practice guidance on the labelling and packaging of medicines The name is defined as comprising the name, strength and pharmaceutical form of the medicine. A number of different scenarios exist and for which different ...
Drug Labeling - StatPearls - NCBI Bookshelf Jun 23, 2022 ... And label and labeling are defined as the following: “(a) ... the content and format of labeling for human prescription drugs and biologics.
15.2 Basic Concepts of Administering Medications - Nursing Skills Special Considerations for Administering Controlled Substances. Controlled substances, also called Scheduled Medications, are kept in a locked system and accounted for using a checks and balance system. Removal of a controlled substance from a medication dispensing system must be verified and documented by a second nurse witness.
4 Controlled Substance Laws and Regulations You Should Know Controlled substance prescriptions have specific requirements. All prescriptions for controlled substances must include the following: 2 Date prescription was issued Prescriber's signature Patient's full name and address Medication name Strength Dosage form Quantity prescribed Directions for use Prescriber's name, address, and registration number.


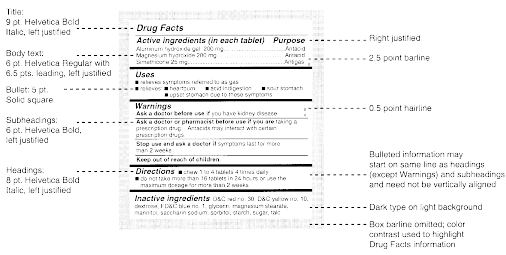

























Post a Comment for "40 explain how controlled substances are identified on their labels"