43 medication labels must include
OTC Labeling Requirements - FindLaw (For example, drug products marketed under the Topical Antifungal Drug Products Monograph 2 should list their active ingredient's purpose as "Antifungal."). Below the "Drug Facts" section, a "Uses" section will list the approved or monograph indications for the drug. The next section of the label is "Warnings." Medication package insert - Wikipedia A package insert is a document included in the package of a medication that provides information about that drug and its use. For prescription medications, the insert is technical, providing information for medical professionals about how to prescribe the drug. Package inserts for prescription drugs often include a separate document called a "patient package insert" with …
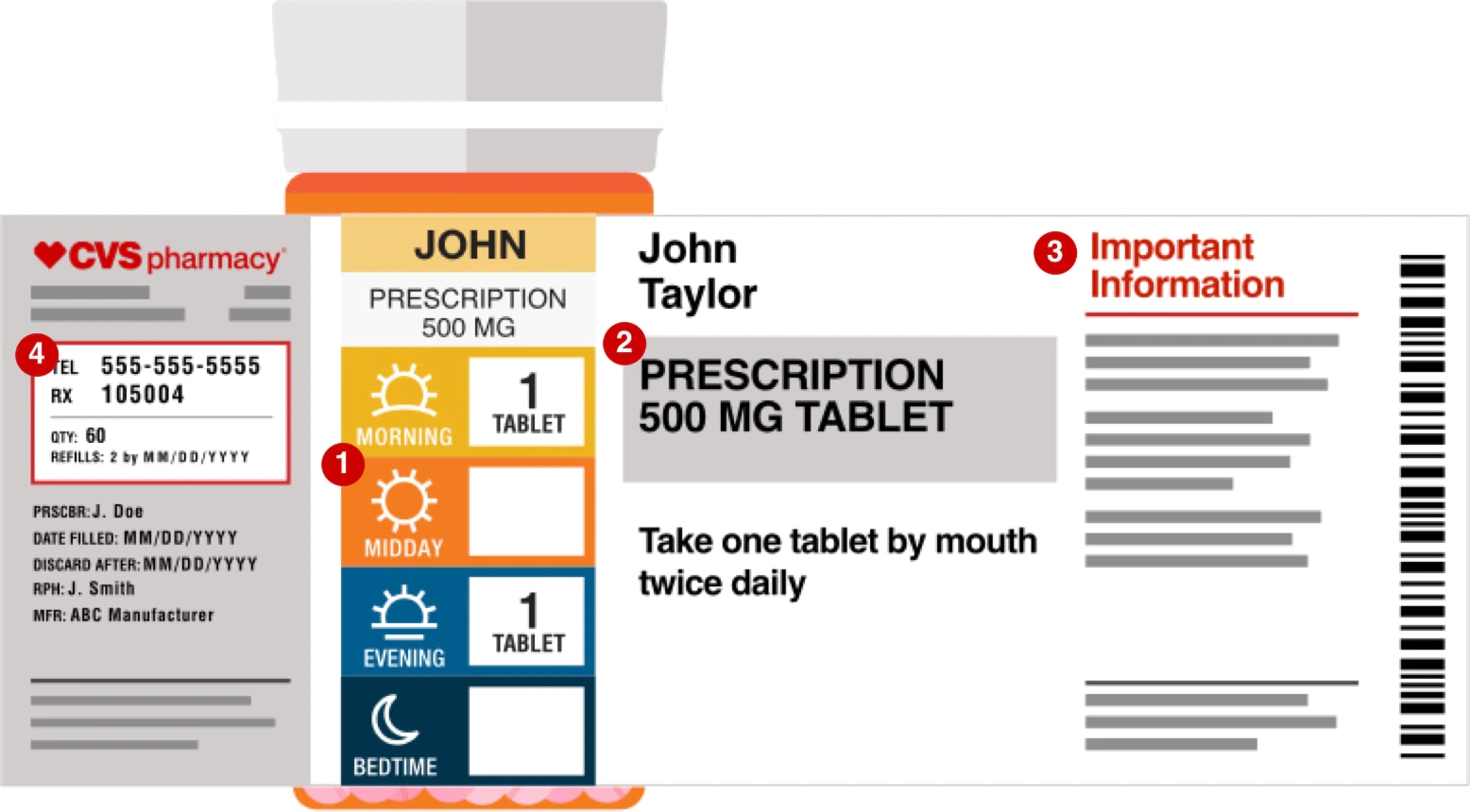
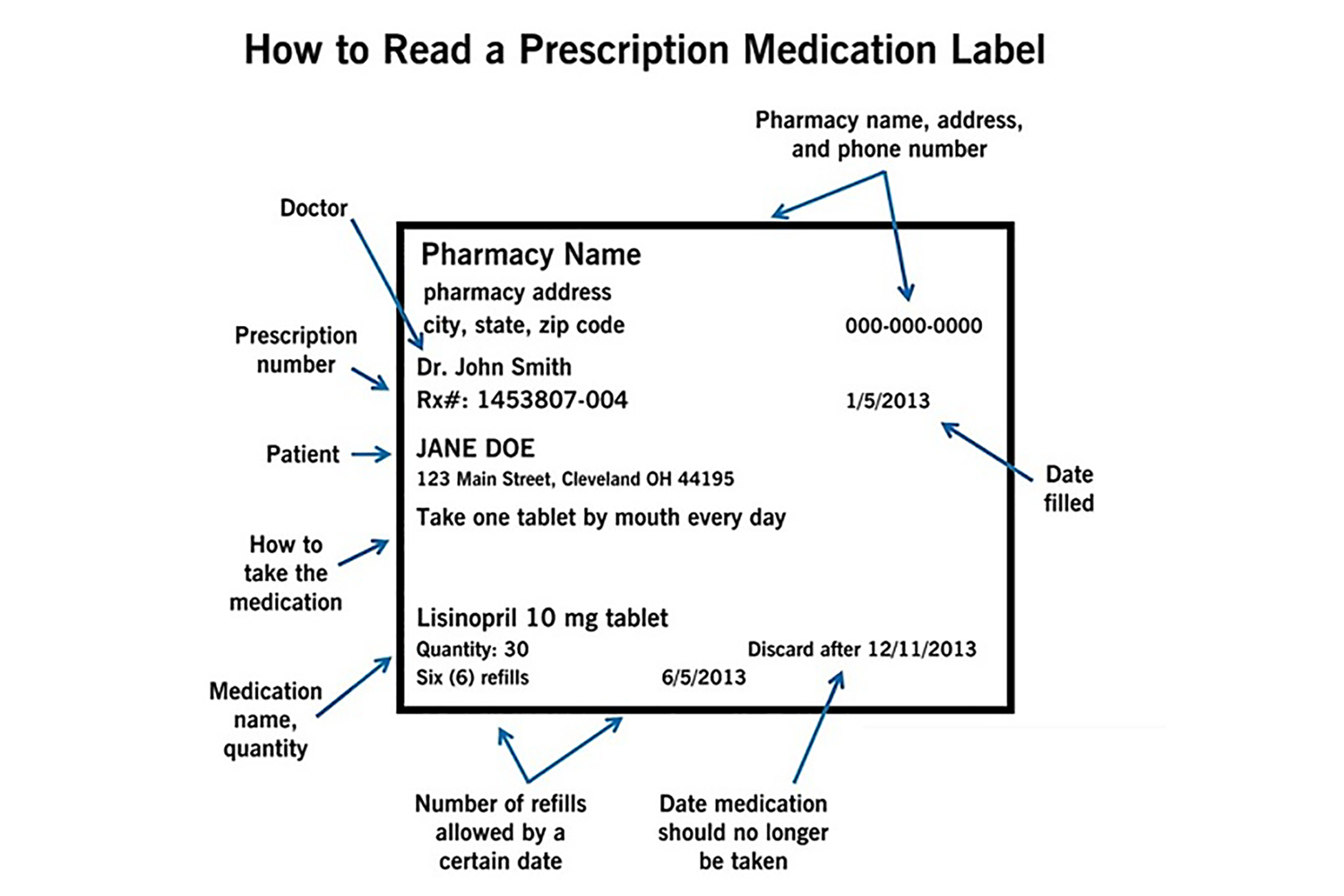
NCBOP - Pharmacist FAQs A. The following information must be on every prescription label: 1. Name and address of the dispensing pharmacy. 2. Serial number of the prescription. 3. Date of the prescription. 4. Name of the prescriber. 5. Name of the patient. 6. Name and strength of the drug. 7.

Medication labels must include
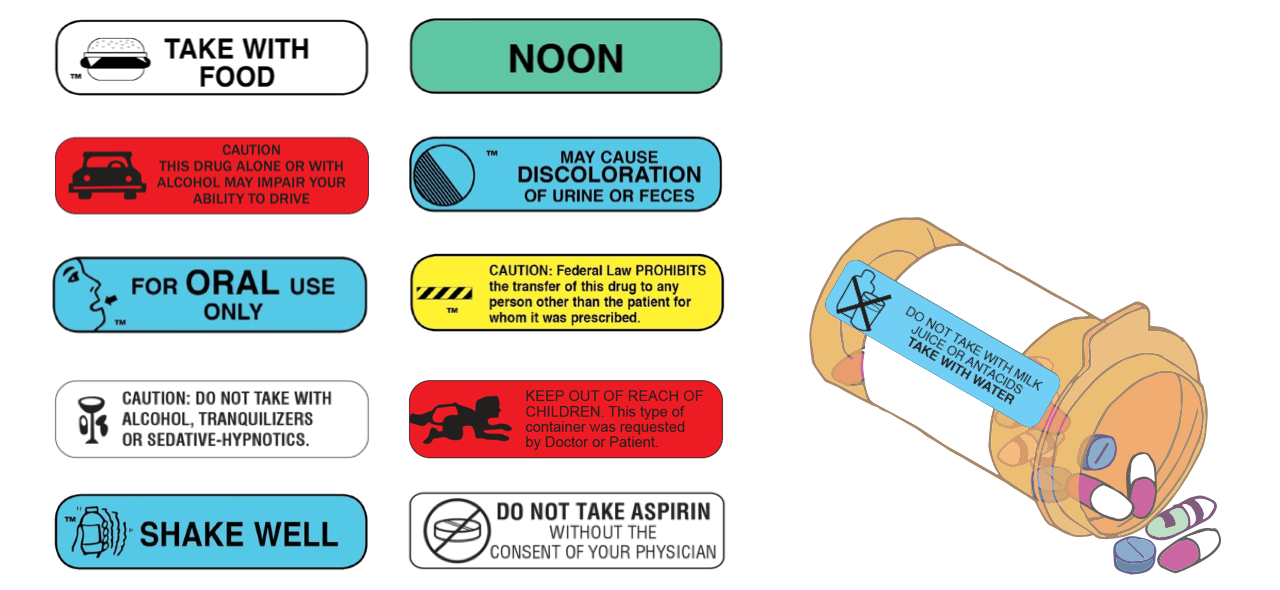
MEDICATION GUIDE SPRAVATO ° Your healthcare provider must monitor you for serious side effects for at least 2 hours after taking SPRAVATO. Your healthcare provider will decide when you are ready to leave the healthcare setting. • Abuse and misuse. There is a risk for abuse and physical and psychological dependence with SPRAVATO treatment. Your healthcare provider should check you for signs of … Pharmacy Prescription Requirements - StatPearls - NCBI Bookshelf There needs to be an understanding by the physician of the mechanism and properties of the medication before prescribing, and the pharmacist must be aware of potential interactions the patient may have with their other medications. Date of issue. Patient's name and address. Patient's date of birth. Clinician name, address, DEA number. Drug name. How to Read Over-the-Counter and Prescription Drug Labels - Drugwatch.com Check the label to make sure your name is on it. If it isn't, talk to the pharmacist. Check the label to make sure you can read and understand the name of the medicine, directions and colored warning stickers on the package. If the letters are too small to read, ask your pharmacist to print it in a larger type.
Medication labels must include. Drug labeling, Information about Drug labeling - FAQs Each product must contain a label with "Supplement Facts" in bold letters onthe front panel. This is the manufacturer's opportunity to identify the product. Below "Supplement Facts," the panel must state the serving size. This isdetermined by the manufacturer with no input from the FDA. What Is a Drug Label? | The Motley Fool Jun 27, 2016 at 11:27PM. A drug label refers to all the printed information included with any dietary supplement, over-the-counter medicine, or prescription drug. They're strictly regulated by the ... Pharmaceutical Labeling: Requirements & Guidelines - CTM Labeling Systems To meet today's FDA regulations, labeling information on drugs must include the following in this order: - Product Name - Drug Facts Table - Active Ingredients - Purpose and Use - Warnings - Directions - Allergic Reactions - Inactive Ingredients PDF Labeling on the Sterile Field: Improve Patient Safety and Ensure Joint ... Labeling must include: Name of medication or solution, strength, date, and time Label one item at a time. Single items must also be labeled.
Safe Labeling Helps Prevent OR Medication Errors - OR Today Label information must include a medication's name and strength as well as amount when medications are mixed (as with antibiotic irrigations, tumescent and heparin solutions, and epinephrine). The unit of measure — percent, grams, milliliters, or units — must be recorded along with the date the medication is prepared. Medication Administration Safety - Patient Safety and Quality Threats to medication safety include miscommunication among health care providers, drug information that is not accessible or up to date, confusing directions, poor technique, inadequate patient information, lack of drug knowledge, incomplete patient medication history, lack of redundant safety checks, lack of evidence-based protocols, and staff assuming roles for which they are not prepared. Legal Requirements of what Must Be on a Prescription Label - MA Pharm.com The labeling required on an OTC product includes: 1. Name of the product 2. Name and address of the manufacturer, packer or distributor 3. Net contents of the package 4. Established name of all active ingredients and the quantity of other ingredients whether active or not 5. The name of any habit-forming drug contained in the preparation 6. A Guide To Veterinary Prescription Label Requirements What Is Required On A Veterinary Prescription Label As shown in the above example, the actual container must include the following information: The name of the veterinary practice, its address, and contact information The veterinarian's name, the patient's name and species, and the client's last name
Safety Considerations for Container Labels and Carton Labeling … The development of labels and labeling is product-specific, and the risk for medication errors differs between products and can be impacted by many factors. Although this guidance provides What's on a prescription label? - Knowledge is the best medicine It uniquely identifies all drug products sold in a dosage form in Canada and is located on the label of prescription and over-the-counter drug products that have been evaluated and authorized for sale in Canada. A DIN uniquely identifies the following product characteristics: manufacturer; product name; active ingredient (s); strength (s) of ... 4. Documenting Medications (MAR). | Aplmed Academy Each medication must be documented at the time of administration. For example, if eight medications are administered the QMAP must initial the MAR eight times indicating that each medication has been administered, refused or unavailable. New order: transcribe new medications on the MAR. 5 Things to Look For in a Prescription Medication Label Here are some Things to Look For in a Prescription Medication Label, from some authorities that set standards for prescription medications - the U.S. Pharmacopeia and the Institute for Safe Medicine Practices. You may be able to find a pharmacy that adheres to some of their suggestions. Drug information is typed in easy-to-read 12-point type ...
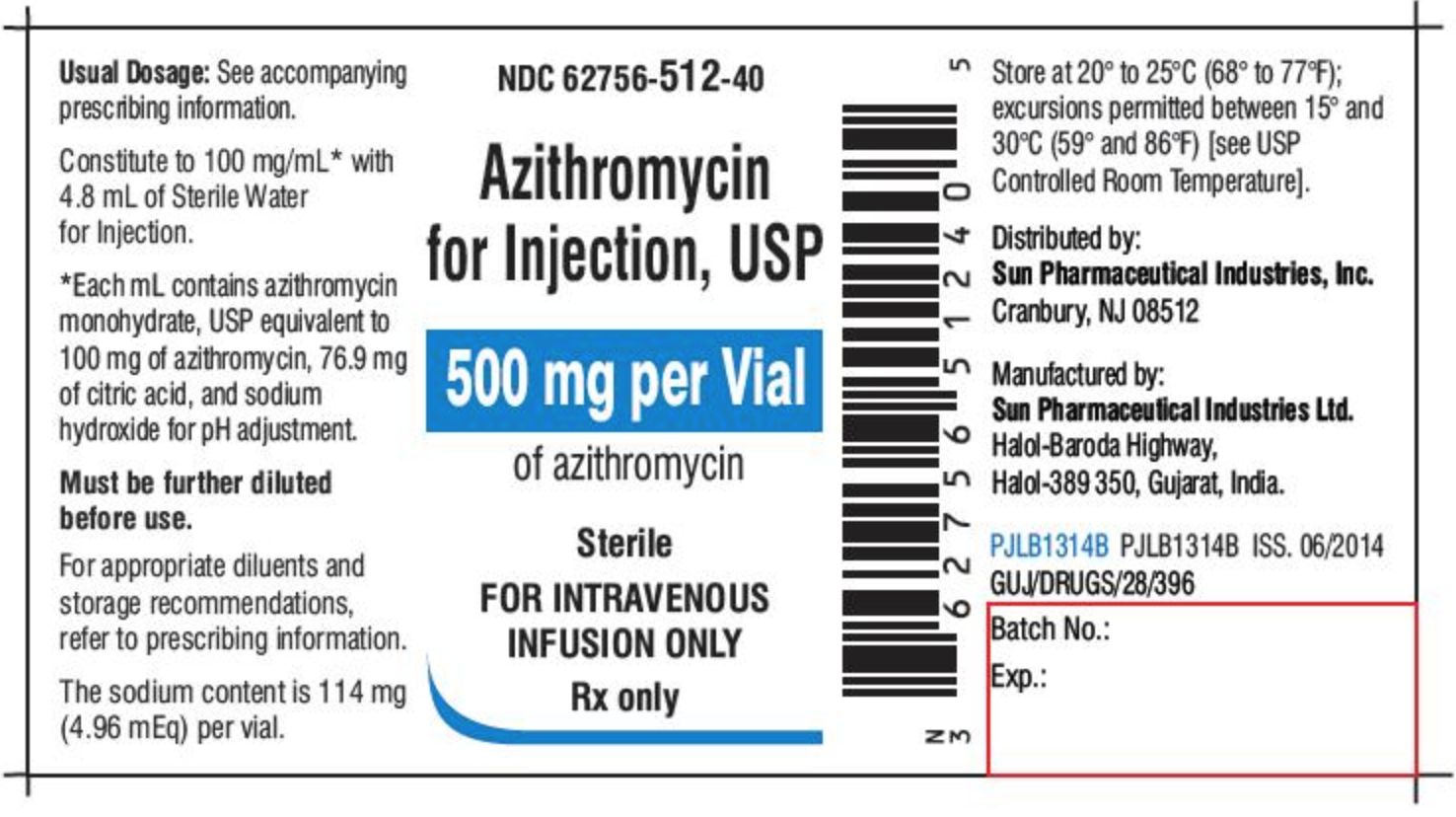
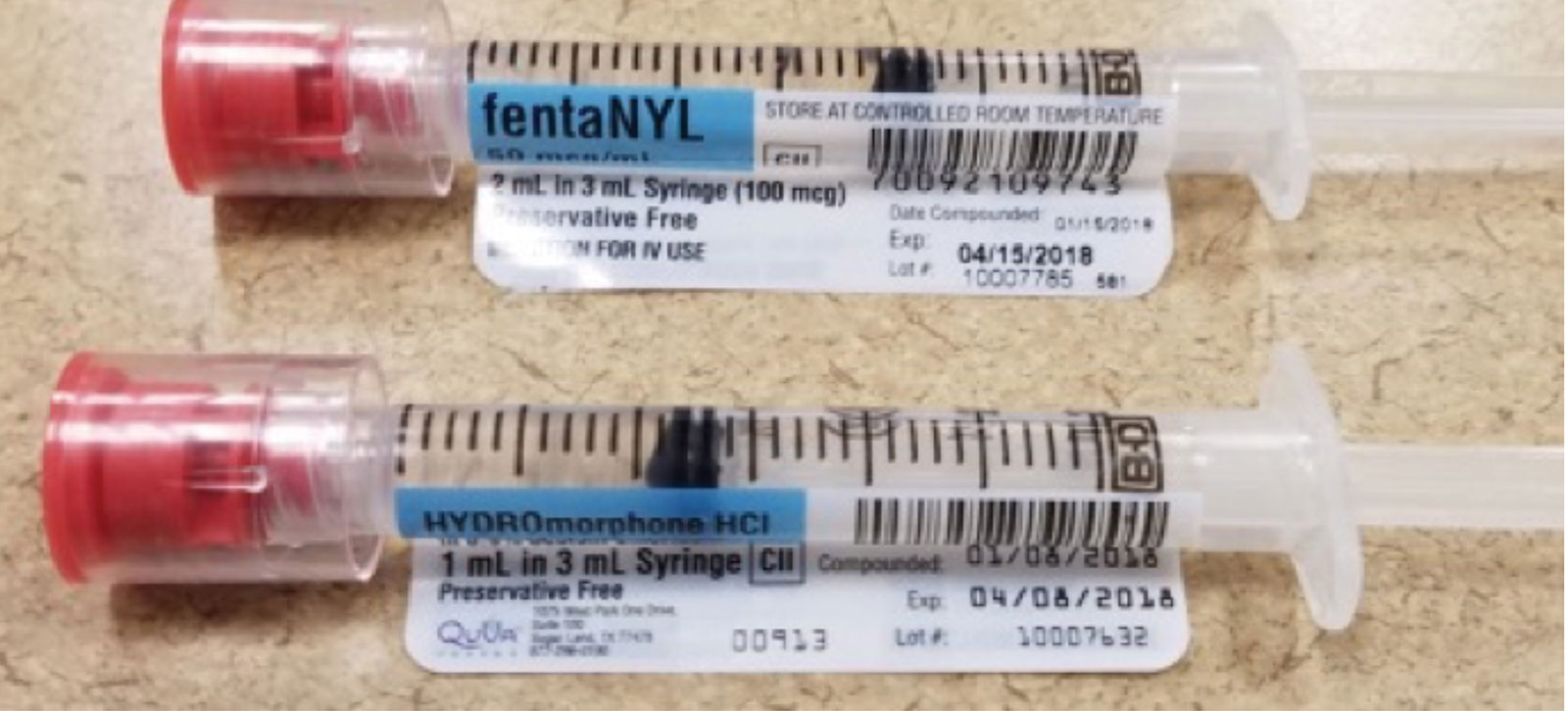
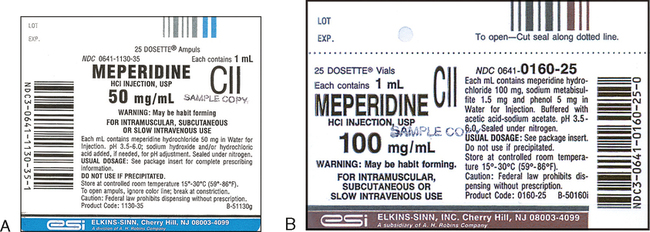
Get ready to assist clients with medication - National Disability … 2. Numbers on medication labels and documents Numbers are everywhere on medication labels and documents. The following medication label and document include examples of information the DSW needs to read and interpret. • Number of tablets • Batch number • Expiry date • Dosage • Age • Temperature • Pregnancy • Barcode ACTIVITY
Medication Study Guide - NCDHHS Labels . 7. Medication Administration Records . 8. Infection Control . You need to read each question thoroughly and choose the best answer. In Section 2, you will find the answers to each of the questions in Section 1. Except for questions 1 through 20, there is an explanation of why the answer is correct. The explanation should help you learn more about your important role with …
The Over-the-Counter Medicine Label: Take a Look | FDA All nonprescription, over-the-counter (OTC) medicine labels have detailed usage and warning information so consumers can properly choose and use the products. Below is an example of what the OTC...
Pharmaceutical Labeling 101: FDA Regulations Guide These include drugs like analgesics, anti-inflammatory agents, antibacterial, anticonvulsants, and others. The substance is used in the diagnosis, mitigation, cure, treatment, or prevention of diseases. This category also includes supplements. The substance is a component of medication but not a part of a medical device.
Over the Counter (OTC) Drug Labels - Poison All OTC drug labels include "Drug Facts", the who, what, how, when, and why of that medicine. The Drug Facts tell you what you need to know to give the right drug, in the rightdose, to the right person, at the right time, in the right way, and for the right purpose.
How to read prescription drug labels - BeMedwise Whenever you are prescribed a medication, you should read and follow the information in the medication's "label" in order to ensure your safety. All prescription medicine containers include information on the label including the patient's name, the name of the medicine, dosage and instructions on how often to take the medicine.
Chapter 5: Prescriptions and Labels Flashcards | Quizlet Drug Labels Regulated by the Food and Drug Administration (FDA), which determines what needs to be on the label Dispensing pharmacist's label must include: Pharmacy name, address, and phone number Dispensing date Dispensing date may differ from the date on the prescription. Rx number, which identifies this unique prescription in the computer system
Pharmacology Chapter 5 (Prescriptions and Labels) - Quizlet Every prescription must include the following:-DATE-Pysicians name, contact info, and DEA number-Patients name, address, and DOB-INSCRIPTION -SIGNATURE ... what other medications include medication labels. over-the-counter (OTC) drugs. OTC labels contain instructions on drug use based on _ and _ age
Drug Labeling - an overview | ScienceDirect Topics The labeling must include the medical indication, effects, and dosage; the route, method, frequency, and duration of administration; and any relevant hazards, contraindications, side effects, and precautions. ... piperine, diosmin, St. John's wort, and grapefruit juice, PGx biomarkers were listed on 366 drug labels and package inserts, of ...
A Primer on Pharmaceutical Label Types and Requirements - Luminer This FDA approved patient labeling typically includes medication guides and instructions for use. Whether or not a given medication must include a medication guide is determined by FDA regulations (usually, if they pose a significant health concern).
Medication - Wikipedia A medication (also called medicament, medicine, ... The drug frequencies are Often expressed as the number of times a drug is used per day (e.g., four times a day). It may include event-related information (e.g., 1 hour before meals, in the morning, at bedtime). or Complimentary to Interval, although equivalent expressions may have different implications (e.g., every 8 hours versus 3 …
Safety Enhancements Every Hospital Must Consider in Wake of … Include IV moderate sedation agents on high-alert medication lists. Include medications commonly used for moderate sedation (e.g., IV midazolam) on the hospital’s list of high-alert medications and implement risk-reduction strategies to prevent errors and patient harm with these medications. For example, administration of IV medications ...
US Food and Drug Administration's Requirements on Content and Format ... With the1979 Labeling Rule, the FDA regulated what information manufacturers must include on a drug label and the way that information was formatted. The rule regulated the label on a drug bottle, as well as the foldable handout that comes with a prescription drug or inside the container of an over-the-counter drug.
How to Label Prescription Medication for Veterinary Patients A label should include the following components: The name of the veterinary practice, its address, and contact information The veterinarian's name, the patient's name and species, and the client's...
FDA's Labeling Resources for Human Prescription Drugs | FDA Human prescription drug labeling (1) contains a summary of the essential scientific information needed for the safe and effective use of the drug; and (2) includes the Prescribing Information,...
Medicines: packaging, labelling and patient information leaflets Labels must include warnings for safe use of the medicine. All products that contain paracetamol must include statutory warnings. Additional warning statements must be included on the packaging of...
Preventing Medication Errors in Hospitals - American Society of … Safe medication practices begin with placing medication safety as anorganizational and departmental priority, and implementing a system that will support these practices. The organization must have a comprehensive program that includes a medication safety leader, key elements in place to provide the structure for safe medication practices, and a
Reading Medication Labels | Basicmedical Key Reading Medication Labels Objectives After reviewing this chapter, you should be able to identify: 1. The trade and generic names of medications 2. The dosage strength of medications 3. The form in which a medication is supplied 4. The total volume of a medication container where indicated 5.
DailyMed - LUPRON DEPOT- leuprolide acetate kit 18.04.2022 · LUPRON DEPOT must be administered under the supervision of a physician. Due to different release characteristics, the dosage strengths are not additive and must be selected based upon the desired dosing schedule. LUPRON DEPOT 7.5 mg for 1-month administration, given as a single intramuscular injection every 4 weeks. LUPRON DEPOT 22.5 mg for 3-month …
FDA Says Drug Labels Must Include Clear Guidance for ... - Healthline Starting June 30, new drug labels will have categories for "Pregnancy," "Lactation," and "Females and Males of Reproductive Potential." "Pregnancy" will include information on ...
How to Read Over-the-Counter and Prescription Drug Labels - Drugwatch.com Check the label to make sure your name is on it. If it isn't, talk to the pharmacist. Check the label to make sure you can read and understand the name of the medicine, directions and colored warning stickers on the package. If the letters are too small to read, ask your pharmacist to print it in a larger type.
Pharmacy Prescription Requirements - StatPearls - NCBI Bookshelf There needs to be an understanding by the physician of the mechanism and properties of the medication before prescribing, and the pharmacist must be aware of potential interactions the patient may have with their other medications. Date of issue. Patient's name and address. Patient's date of birth. Clinician name, address, DEA number. Drug name.
MEDICATION GUIDE SPRAVATO ° Your healthcare provider must monitor you for serious side effects for at least 2 hours after taking SPRAVATO. Your healthcare provider will decide when you are ready to leave the healthcare setting. • Abuse and misuse. There is a risk for abuse and physical and psychological dependence with SPRAVATO treatment. Your healthcare provider should check you for signs of …



























Post a Comment for "43 medication labels must include"